 In a neutral atom, the atomic and ionic radius are the same, but many elements exist as anions or cations. Ionic radius is the distance from the nucleus to the outer edge of the electron cloud of an ion. Why does ionic radius increase down a group?ĭown a group, the number of energy levels (n) increases, so there is a greater distance between the nucleus and the outermost orbital. Therefore, ionic radii increase down a group as more shells are added (per period). In a group, all the ions have the same charge as they have the same valency (that is, the same number of valence electrons on the highest energy level sub-orbital). 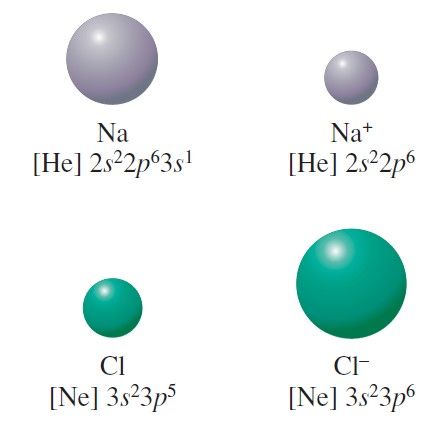
Does ionic radius increase down the group? Take the humble carbon atom as an example: in most organic molecules a covalently-bonded carbon atom is around 1.5 Ångstroms in diameter (1 Ångstrom unit = 0.1 nanometres = 10-10 metres) but the same atom in an ionic crystal appears much smaller: around 0.6 Ångstroms. The radius of the ion depends on the effective nuclear charge, more the charge, more heavily the electrons are attracted towards the center and lesser in the size. : Bond length of `Cl – Cl ” in ” Cl_(2)` molecule is 198 pm. Solution : Covalent Radius: “When two atoms are bound together by a single bond in a covalent molecule distance between two atoms from centre is known as covalent radius.”Į.g. In principle, the sum of the two covalent radii should equal the covalent bond length between two atoms, R(AB) = r(A) + r(B). It is usually measured either in picometres (pm) or angstroms (Å), with 1 Å = 100 pm. The covalent radius, rcov, is a measure of the size of an atom that forms part of one covalent bond. In non-metals, the atomic radius is smaller than the ionic radius. This will create a larger positive charge in the nucleus causing the electron cloud to come closer to the nucleus. Because they lose electrons for the formation of octets. In metals, the atomic radius is larger than the ionic radius. Why ionic radius is greater than atomic radius? Ions are formed when an atom loses or gains electrons. 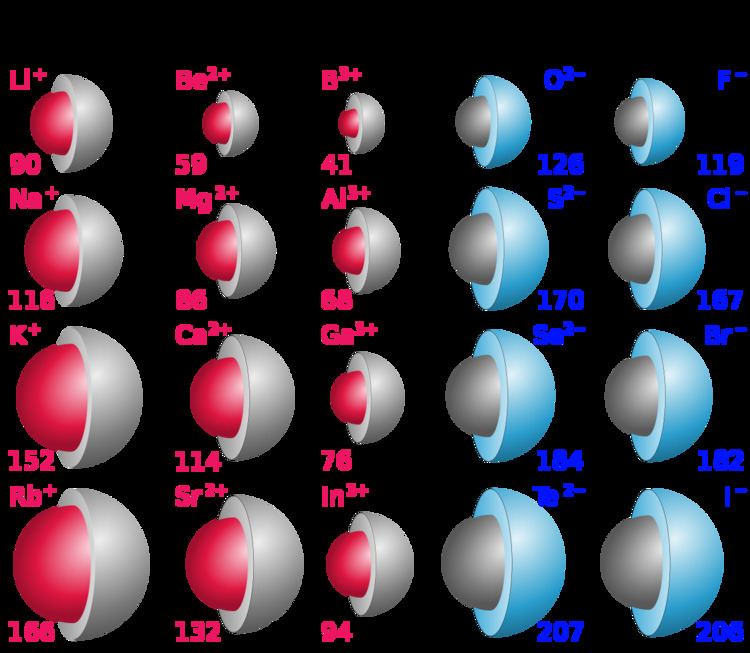
What is Ionic Radius? Ionic radius is the distance from the nucleus of an ion up to which it has an influence on its electron cloud. Metal atoms lose electrons to become ions, so their ionic radius is smaller than their atomic radius. Electrons are the outermost part of atoms, so are directly responsible for the radius. When an atom becomes an ion, it gains or loses electrons. What is the difference between ionic radius and metallic radius? Ionic radius increases down the group ,in the following option strontium is having the largest ionic radius as the elements arranged in periodic table is Berilyium, Magnesium, Calcium and Strontium. Although neither atoms nor ions have sharp boundaries, they are treated as if they were hard spheres with radii such that the sum of ionic radii of the cation and anion gives the distance between the ions in a crystal lattice. Ionic radius, rion, is the radius of a monatomic ion in an ionic crystal structure. Solution : Ionic radii is defined as distance from the centre of nuclei up to which influence of nucleon can be felt.Ĭovalent radii is half of the distance between centre of nuclei of two covalently bonded atoms. Ionic radius is a measure of an atoms ion in a crystal lattice and which is a half distance between two ions that are barely touching each other. 
24 Why is the ionic radius of Na+ less than F? What is atomic radius and ionic radius?Ītomic radius is defined as a distance from the center of the nucleus to the outermost shell containing the electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
